T-2 Mycotoxin (T-2)
Background and History:
- T-2 is a trichothecene mycotoxin that is naturally occurring byproduct of Fusarium fungus. T-2 mycotoxin is the only toxin produced by fungi known to be used as a biological weapon. Unlike other biological toxins, T-2 mycotoxin is a potent dermal irritant and can be absorbed through intact skin causing systemic toxicity [1].
- Large amounts of the toxin are needed for the lethal dose. The LD50 of T-2 toxin is approximately 1mg/kg. The toxin is relatively insoluble in water and highly soluble in ethanol, methanol, and propylene glycol.
- The mycotoxins are extremely stable proteins that are resistant to heat and ultraviolet light. To inactivate the toxin it needs to be heated to 500F for 30 minutes or exposure to sodium hypochlorite [3].
- T-2 toxin was first discovered in 1931 by Ukrainian veterinarians who reported a unique disease in horses. They noted lip edema, oral necrosis, and it progressed to coagulopathy, neurologic compromise, and death. They named the disease alimentary toxic aleukia (ATA) [4].
- It was realized in Russia during World Was II that it could be used as a biological weapon when civilians consumed wheat contaminated with Fusarium fungi. The victims contracted ATA. Allegations surrounding the use of T-2 toxin as a warfare agent remains controversial. Eyewitness and victim accounts reported seeing a low-flying aircraft that dropped a yellow, oily liquid on victims in Russia. Allegedly, T-2 has been used in military conflicts in Laos (1975-1981), Kampuchea (1979-1981), and Afghanistan (1979-1981) with over 10,000 combined deaths.
- Transmission of the toxin can be via food, water sources, droplets, and aerosols. The toxins can be absorbed topically, orally, and by inhalation. It is thought to be 400 times more potent than sulfur mustard as a skin irritant and blistering agent.
- Witnesses have reported clouds of yellow, blue, or green aerosols. If the toxin is ingested by contaminated foods it has no taste, no foul odor, and no suspicion of tainted food [4].
Mechanism:
- Trichothecene mycotoxins are cytotoxic and immunosuppressive.
- They inhibit protein and nucleic acid synthesis. This causes apoptosis through robotic stress and reactive oxygen species-mediated mitochondrial pathway (Please see image above for graphic of mechanism).
- It is most pronounced in actively proliferating skin cells, GI cells, and bone marrow.
- It is also thought to disrupt DNA polymerases, monoamine oxidase, and other proteins involved with coagulation.
- T-2 toxin is rapidly metabolized to HT-2, T2-triol, and T-2 tetaol and many other metabolites within hours of exposure [2]
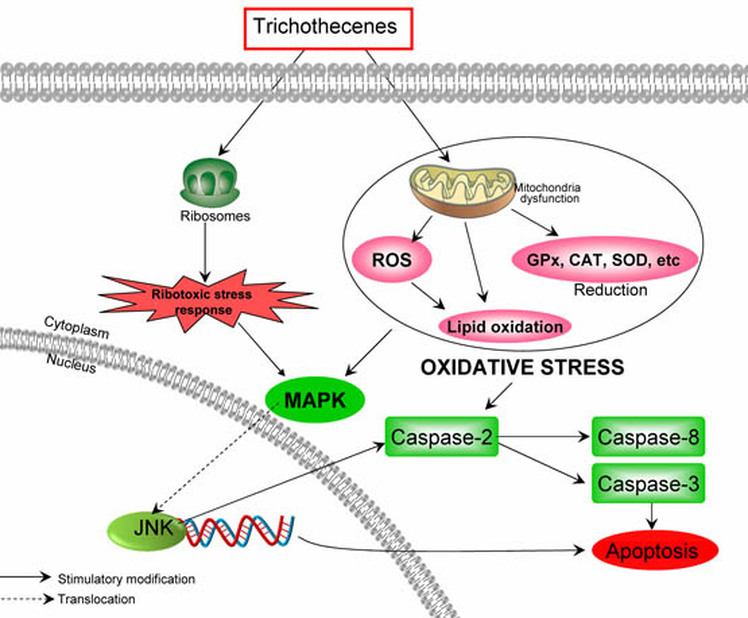
Fig 1: Pathway of Trichothecenes into cells. Reactive oxygen species occur through mitochondria dysfunction. Ribosomes dysfunction occurs. MAPK is disrupted and apoptosis occurs.
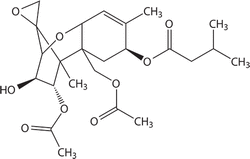
Fig 2: Molecular structure of T-2 Mycotoxin.
Signs and Symptoms:
Symptoms may manifest within a few seconds up to a few hours depending on the dose of exposure and the following signs and symptoms may occur [4]:
• Neurologic: mild ataxia (difficulty walking), severe dizziness, hypothermia
• Ocular: tearing, pain, conjunctivitis, and blurred vision.
• Nasal: rhinorrhea, sneezing, epistaxis (bleeding from nose)
• Oral: pain/buring, blood-tinged saliva and sputum
• Respiratory: cough, dyspnea, wheezing, hemoptysis (coughing up blood)
• Cardiovascular: tachycardia, DIC
• GI: N/V, watery diarrhea, abdominal cramps,
• Dermal: erythema, tenderness, blistering, bullous lesions, necrosis, sloughing of dermal layer.
The long term symptoms from exposure to the toxin are experienced in four different stages:
Symptoms may manifest within a few seconds up to a few hours depending on the dose of exposure and the following signs and symptoms may occur [4]:
• Neurologic: mild ataxia (difficulty walking), severe dizziness, hypothermia
• Ocular: tearing, pain, conjunctivitis, and blurred vision.
• Nasal: rhinorrhea, sneezing, epistaxis (bleeding from nose)
• Oral: pain/buring, blood-tinged saliva and sputum
• Respiratory: cough, dyspnea, wheezing, hemoptysis (coughing up blood)
• Cardiovascular: tachycardia, DIC
• GI: N/V, watery diarrhea, abdominal cramps,
• Dermal: erythema, tenderness, blistering, bullous lesions, necrosis, sloughing of dermal layer.
The long term symptoms from exposure to the toxin are experienced in four different stages:
- Stage 1: This stage may be seen in the emergency department. This stage results from the acute injury to the exposed cells and tissue. The symptoms reflect the route of toxin exposure.
- Stage 2: This stage occurs weeks after the exposure. Insult to the bone marrow initially produces a transient lymphocytosis. This is soon followed by bone marrow suppression due to the antimitotic effects of T-2 toxin. The result is significant leukopenia, granulocytopenia, and thrombocytopenia.
- Stage 3: This stage also occurs weeks after the exposure and is considered the hemorrhagic stage. The patient exhibits petechial hemorrhages, especially of the mucosal areas of the nasopharynx and oropharynx. These lesions develop to form ulcerated and necrotic lesions, which can result in significant bleeding from the esophagus and the gastrointestinal tract. Moreover, the edema that accompanies the mucosal injury may threaten the airway. Also, severe coagulopathy may occur. During this stage, the patient is at a higher risk for sepsis because the immune system is significantly compromised.
- Stage 4: During the recovery phase, the necrotic lesions heal and the bone marrow recovers [4].
Treatment:
- Remove all of the patient’s clothing. Clean and scrub the skin with soap and water.
- If the affected area has been decontaminated within 6 hours after exposure that can remove 80-98% of the toxin preventing skin lesions and death.
- Health care providers need to use universal precautions.
- Patient’s with high exposures much be monitored for any problems breathing, airway, and circulation.
- Keep clothing contained to avoid cross contamination.
- Activated charcoal may be given for ingested exposures within 1 hour post-exposure [4].
Citations:
[1] Fang, H., Wu, Y., Guo, J., Rong, J., Ma, L., Zhao, Z., . . . Peng, S. (2012). T-2 toxin induces apoptosis in differentiated murine embryonic stem cells through reactive oxygen species-mediated mitochondrial pathway. Apoptosis, 17(8), 895-907.
[2] Kuca, K., Dohnal, V., Jezkova, A., & Jun, D. (2008). Metabolic Pathways Of T-2 Toxin. Current Drug Metabolism, 77-82.
[3] Liu, J., Guo, X., & Wu, S. (2014). The role of mitochondria in T-2 toxin-induced human chondrocytes apoptosis. PLOS One, 9(9).
[4] Park, C, MD. CBRNE - T-2 Mycotoxins . Retrieved April 22, 2015, from http://emedicine.medscape.com/article/830892-overview
[1] Fang, H., Wu, Y., Guo, J., Rong, J., Ma, L., Zhao, Z., . . . Peng, S. (2012). T-2 toxin induces apoptosis in differentiated murine embryonic stem cells through reactive oxygen species-mediated mitochondrial pathway. Apoptosis, 17(8), 895-907.
[2] Kuca, K., Dohnal, V., Jezkova, A., & Jun, D. (2008). Metabolic Pathways Of T-2 Toxin. Current Drug Metabolism, 77-82.
[3] Liu, J., Guo, X., & Wu, S. (2014). The role of mitochondria in T-2 toxin-induced human chondrocytes apoptosis. PLOS One, 9(9).
[4] Park, C, MD. CBRNE - T-2 Mycotoxins . Retrieved April 22, 2015, from http://emedicine.medscape.com/article/830892-overview
